The Essentials of Surface Wipe Sampling for Pharmacies
Latest posts by Eagle Analytical (see all)
- Eagle New Year Party & Eagle Employee Of The Year Award Ceremony, 01/13 - March 11, 2024
- HRT Functional Medicine Symposium, 02/15 – 02/17 - March 11, 2024
- APhA, 03/22 – 03/25 - March 11, 2024





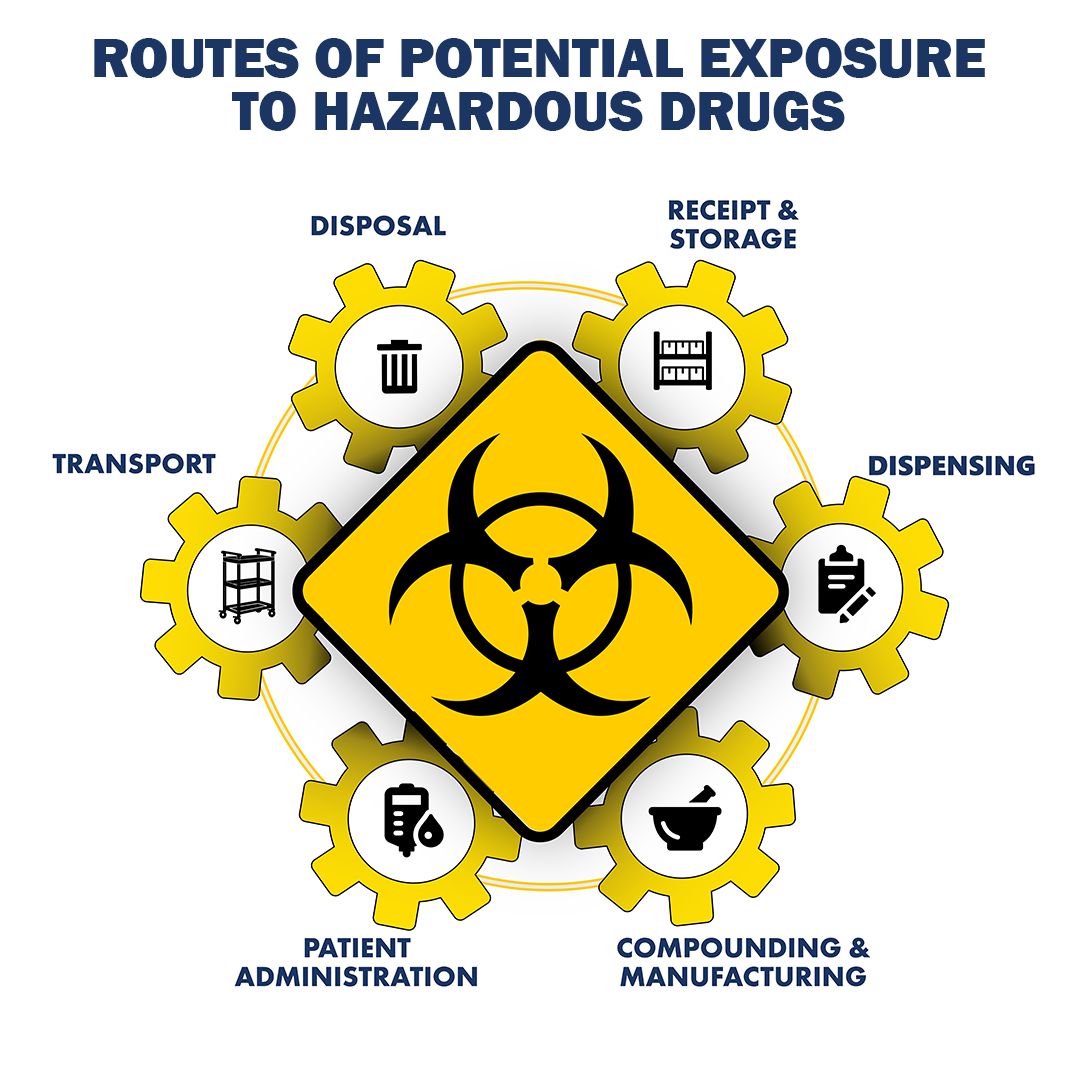
Any organization, both sterile and nonsterile, handling hazardous drugs (HDs) should be tested for surface contamination. This best practice helps to evaluate the environment for chemical residue, minimize the risk of drug exposure by healthcare personnel and patients, and avoid cross-contamination of the product. Surface wipe sampling is encouraged in USP 800 standards and required as part of the cleaning validation program for all CGMP facilities under Title 21 Part 211 of the Code of Federal Regulations (CFR) requirements.
Thank you for reading this post, don't forget to subscribe!According to USP 800, wipe sampling should be performed initially, to establish a baseline of surface contamination, then no less than every six months to verify containment. However, more frequent sampling should be considered after data has been evaluated and a risk assessment has been performed.
Typically, the locations with higher activities are areas of concern and should be tested. The receipt and storage, dispensing, compounding, administration disposal, and transport are common spaces for evaluation.
Learn More About Surface Sampling | SurfaceShield Wipe Sampling and Cleaning Verification Kit