An informational series created to assist you in understanding and navigating EagleTrax’s Shield Analysis statistical process control charts. Save this page to your favorites for quick access to new Shield Analysis videos, how-to instructions, pdfs, and related content.
Process Control Charts -
Harness the Power of Trending
The new Shield Analysis feature of EagleTrax provides a comprehensive and retrospective review of personnel and environmental monitoring data as well as critical quality attributes of the compounded preparation, enabling users to analyze trends over time. By examining data points collectively, Shield Analysis can assist in interpreting the overall state of microbial and product quality control. The process control charts that are generated through Shield Analysis may serve various purposes, including but not limited to the proactive identification of adverse trends, review of data through graphical representation, monitoring of microbial flora and evaluation of seasonal trends, evaluation of the cleaning and disinfection program, satisfy regulatory requirements for trending and annual product review, and identification of problem areas.
Process Control Chart Types
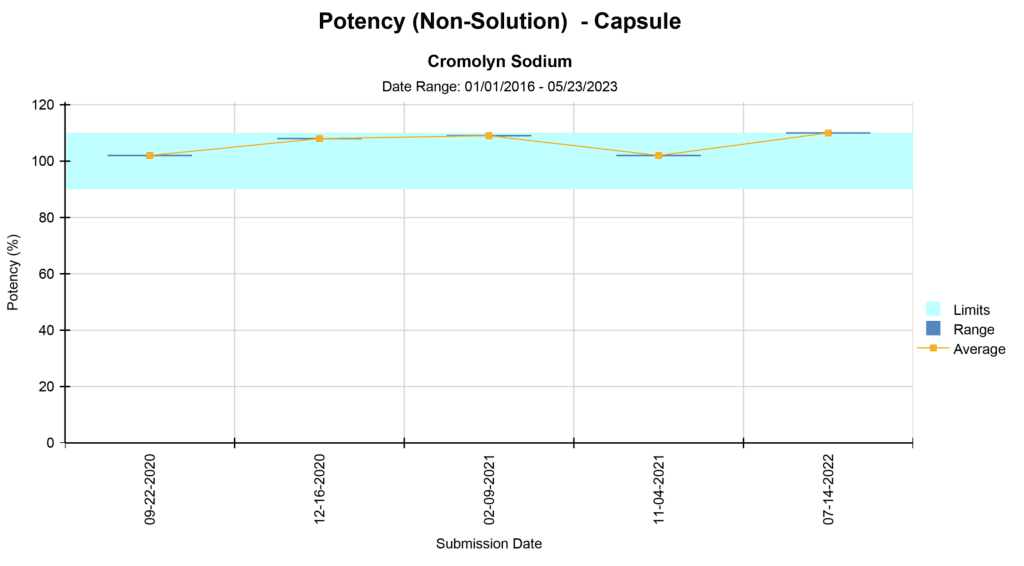
We offer various types of reports, including Microbial Identification, Potency Average, Potency Multiple Analytes, Potency Trends, SCANRDI®, USP <71> Sterility, USP <85> Bacterial Endotoxin, and USP <800> Hazardous Drugs reporting. Here are some examples of reports for your reference.



Shield Analysis Informational Series
Creating a Sample Submission for Environmental Monitoring Sampling
To ensure the accurate and proper submission of test samples, we suggest standardizing the submission process and filling the following fields with the below information that will facilitate future trend analysis over time.
Let Our Experience Work For You
Cutting-Edge Our Laboratories
Eagle’s cutting-edge 80,000-square-foot facility and laboratory are equipped with advanced technologies and specialized segregated laboratory spaces to meet the diverse needs of our clients. This behind-the-scenes video provides you with an opportunity to witness testing while touring our laboratory.
