The World’s Fastest Sterility Test
A Faster Way to Comply
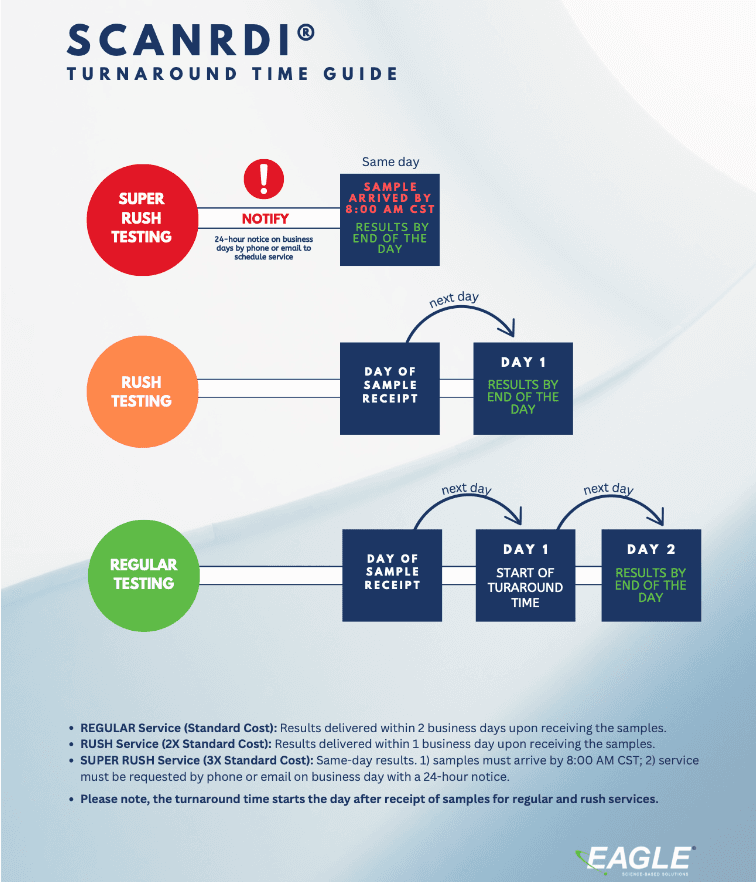
The ScanRDI® is a unique alternative technology that enables Eagle to deliver sterility testing results in 1 or 2 business days†, allowing sterile compounding facilities to release product faster than when utilizing the compendial USP <71> method. ScanRDI® has revolutionized rapid microbial detection and its combination of speed and sensitivity remains unrivaled to this day(a). Additionally, ScanRDI® fulfills the method suitability requirement as outlined in USP <1223>.
Time Savings Equal Financial Savings
Results in 1 Business Day†
Now there is a faster way to comply with USP <797> and ensure your patients are receiving quality compounds. ScanRDI® is a non-growth-based technology sensitive enough to detect a single bacterial, yeast, or mold contaminant in a matter of hours. Even spores, stressed and fastidious organisms are detected in minutes, offering an extraordinarily rapid alternative to the traditional 14-day sterility test(a).
Maintain Compliance Accepted Alternative
The ScanRDI® is an accepted alternative to the official compendial sterility test method.**
Detects Viable Microbial Cells
ScanRDI® rapidly detects viable microbial cells without the need for growth or cell manipulation.
Forgoes the Extended Incubation Period
This forgoes the extended incubation period included in the USP Test Method, so results are ready in 1 Business day.†
Featuring Eagle
bioMerieux's "Ask The Experts" Series
As an industry leader in analytical testing and consulting services for 503A compounding pharmacies, 503B outsourcing facilities, and pharmaceuticals, Eagle Analytical embraces a philosophy called the ‘Eagle Shield’ to help customers get product to market faster, safely and with full compliance.
The Limitations of USP
In order to fully comply with USP regulations, a facility producing sterile product must develop a formal quality assurance (QA) program. One of the characteristics of a QA program includes routine testing of sterile preparations. However, the USP <71> sterility test method presents the following limitations:
14 Days of Incubation
The compendial method of testing takes 14 to 18 days to complete, due to the incubation period required in the testing procedure.
Decreased Patient Use Time
As a result, a sterile compound with a standard 30-day expiry loses approximately half of its shelf life due to sterility testing.
Patented Method
Now Serving Rapid Sterility Tests on Oils with ScanRDI®
Eagle has developed and patented a method that allows us to test your oil-based preparations using the ScanRDI® Sterility Test. Eagle President and CEO Ross Caputo, Ph.D., explains this breakthrough discovery in this video.

Get Started Today
Got Sterility Testing Needs?
Call the Eagle Client Care Team at (800) 745-8916 to discuss your operation’s specific needs and answer any questions!

