Operations Services —
Calibration, Certification, Qualification, and Facility Design Review
BUILDING BETTER
Our engineers know that you rely on your equipment to perform as expected. These instruments operate as a safeguard for the quality of your products and for the well-being of your employees. To ensure your equipment is operating properly, international standards recommend that most instruments be tested and calibrated. An example of this is after an Installation Qualification, Operational Qualification, and Performance Qualification (IQ/OQ/PQ) are completed, equipment is routinely inspected, calibrated, and checked to ensure proper performance. Eagles engineers will ensure your systems are performing as designed. With Eagle, you will feel confident that your equipment is operating as intended.
Certification
- Laminar Airflow Workspaces (LAFW)
- Biological Safety Cabinets (BSC)
- Powder Containment Hoods
- Fume Hoods (coming soon)
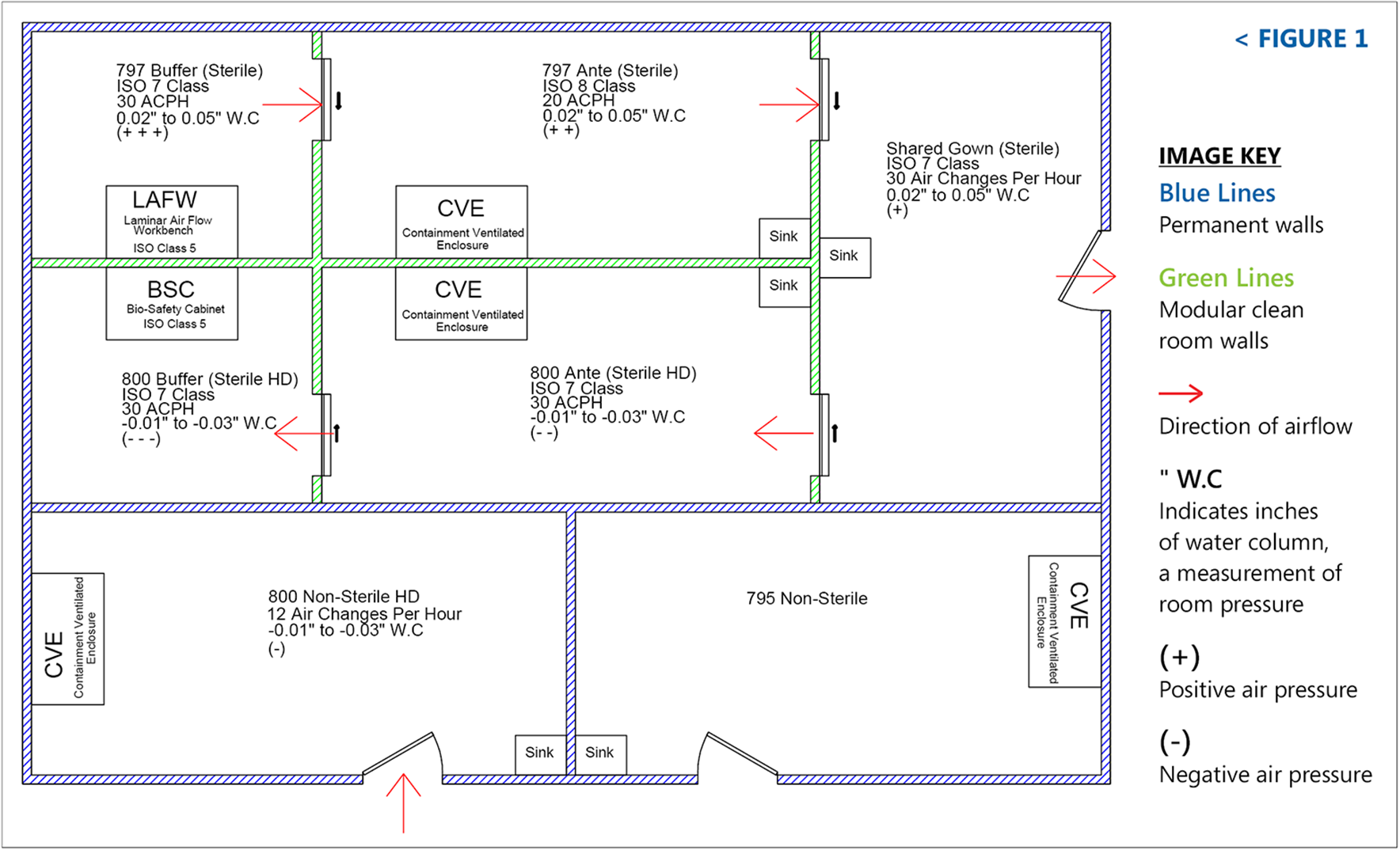
- Full Cleanroom Certification to <797> and CETA Standards to include:
- HEPA Filter Integrity Testing (Leak Test)
- Differential Pressure Measurement
- Air Change Per Hour Measurement and Calculation
- Non-Viable Air Sampling (Particle Counts)
- Viable Air and Surface Sampling (Environmental Monitoring)
- Static Air Visualization (Smoke) Study
- Dynamic Air Visualization (Smoke) Study with Video and Report
Calibration
- Balances
- Temperature Sensors and Thermometers
- Relative Humidity Sensors
- Differential Pressure Sensors (digital and magnehelic)
- Controlled Temperature Units
- Pipettes (coming soon)
Qualification ¹
- Installation/Operational Qualification and Performance Qualification:
- Autoclave – Sterilization Cycle Validation
- Dry-Heat Oven – Depyrogenation Cycle Validation
- Incubators
- Refrigerators
- Freezers
- Stability Chambers
Need help?
Complete the form to request an initial consultation so that we may discuss your operation’s specific needs.
FACILITY DESIGN AND REVIEW
Our engineering team will guide you through the building design and construction process, whether it be at startup or transition, to ensure that your facility is in compliance with the standards that govern your operations, including:
- Current Good Manufacturing Practice (CGMP)
- ISO 14644 Cleanrooms and Associated Controlled Environments
- USP <795> Pharmaceutical Compounding – Nonsterile Preparations
- USP <797> Pharmaceutical Compounding – Sterile Preparations
- USP <800> Hazardous Drugs – Handling in the Healthcare Settings
We can assist you in determining the optimal layout of your space to maximize efficiency and workflow all while keeping growth in mind.
Note:
¹ Other equipment is available upon request.
Operations Team
Calibration, certification, qualification, and facility design review services are offered by Eagle’s Operations Department. The operations team consents of dedicated engineering and logistics professionals who are committed to providing expert and excellent care to our customers.

Jeff

Chris

John

Terreal

James

Rochard

