USP Revisions
Cleanroom Certification
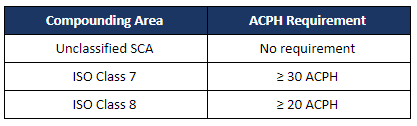
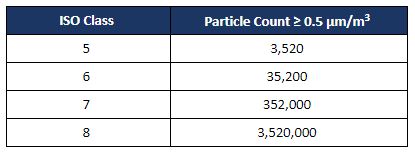
Despite revisions to the sterile compounding chapter, USP <797> that became official on November 1, 2023, the requirements for cleanroom certification remain consistent with the 2008 standards.
Classified areas and engineering controls must be certified to meet the requirements for the following test: