
Environmental Monitoring & Why it Matters: Part 2
Read Previous Article: Environmental Monitoring and Why it Matters – Part 1
Thank you for reading this post, don't forget to subscribe!The Importance of Trending and Analyzing Data
Chapter <797> of the United States Pharmacopeia (USP) outlines the operational and facility requirements for compounding sterile drugs. One specific area of the quality assurance program prescribed is the collection and analysis of environmental monitoring data through air and surface sampling.
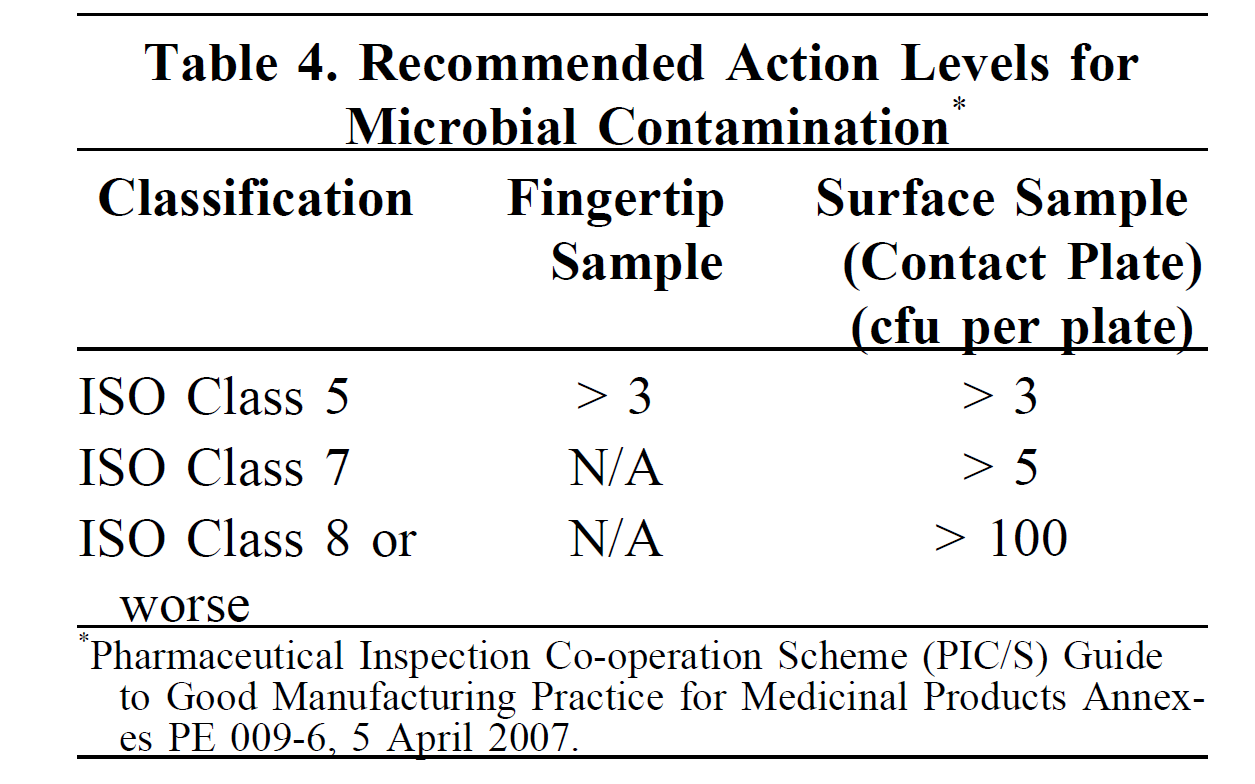
Samples are collected, incubated on growth media, and then examined for the development of microbial growth (typically shown as colonies on solid media). The count of colonies is reported as colony forming units (CFUs). The number of CFUs recovered should not exceed established excursion levels. USP <797> provides initial recommended maximum levels.
| For example, work-surface-microbial-contamination action levels are currently set as >3, >5, and >100 CFU/plate in the ISO 5, 7, and 8 areas, respectively (Table 4 in chapter <797> of USP 40). |
Making this Data Useful
While most compounding pharmacies assess whether the number of CFUs recovered are within these limits, compounders commonly do not track, trend, or analyze these data past this assessment. This is where the implementation of an environmental monitoring program often falls short: data collected from environmental monitoring is most useful when it is trended over time and reviewed frequently in order to detect the development of dynamic negative trends. Data from an environmental monitoring program is trended and can be analyzed dynamically to provide critical indicators about the state of control of the sterile compounding environment.
Staying Ahead of Microbial Contamination
By tracking and trending environmental monitoring data over time, compounders may predict whether the environment will deteriorate from a proper state of control and then are able to implement changes. For example, data that shows negative trends in the level of environmental quality can indicate a loss of control over the microbiological quality of the facilities. This early warning of a negative development allows compounders to react proactively and implement corrective actions before product safety is compromised.
Trending environmental data, therefore, becomes a powerful tool that both protects the quality of the product and makes good business sense because it is less costly to prevent a problem than to react to one that has already occurred.
In the next article, we’ll discuss Using the Right Metric to analyze data collected from an environmental monitoring program, per USP <1116>.
Need Help With Your Environmental Monitoring Program?
Eagle consultants are helping compounding facilities implement successful quality systems. Our team has over 200 years of combined experience in the FDA-regulated pharmaceutical industry and can provide guidance in establishing the systems and processes that will help your facility meet and exceed regulatory expectations. Follow the link below to learn more about our consulting services.
- Environmental Monitoring & Why it Matters: Part 3 - February 26, 2020
- Environmental Monitoring & Why it Matters: Part 2 - February 12, 2020
- Environmental Monitoring & Why it Matters: Part 1 - February 5, 2020




