Description

| SurfaceShield Wipe Sampling and Cleaning Verification Kit Includes¹ |
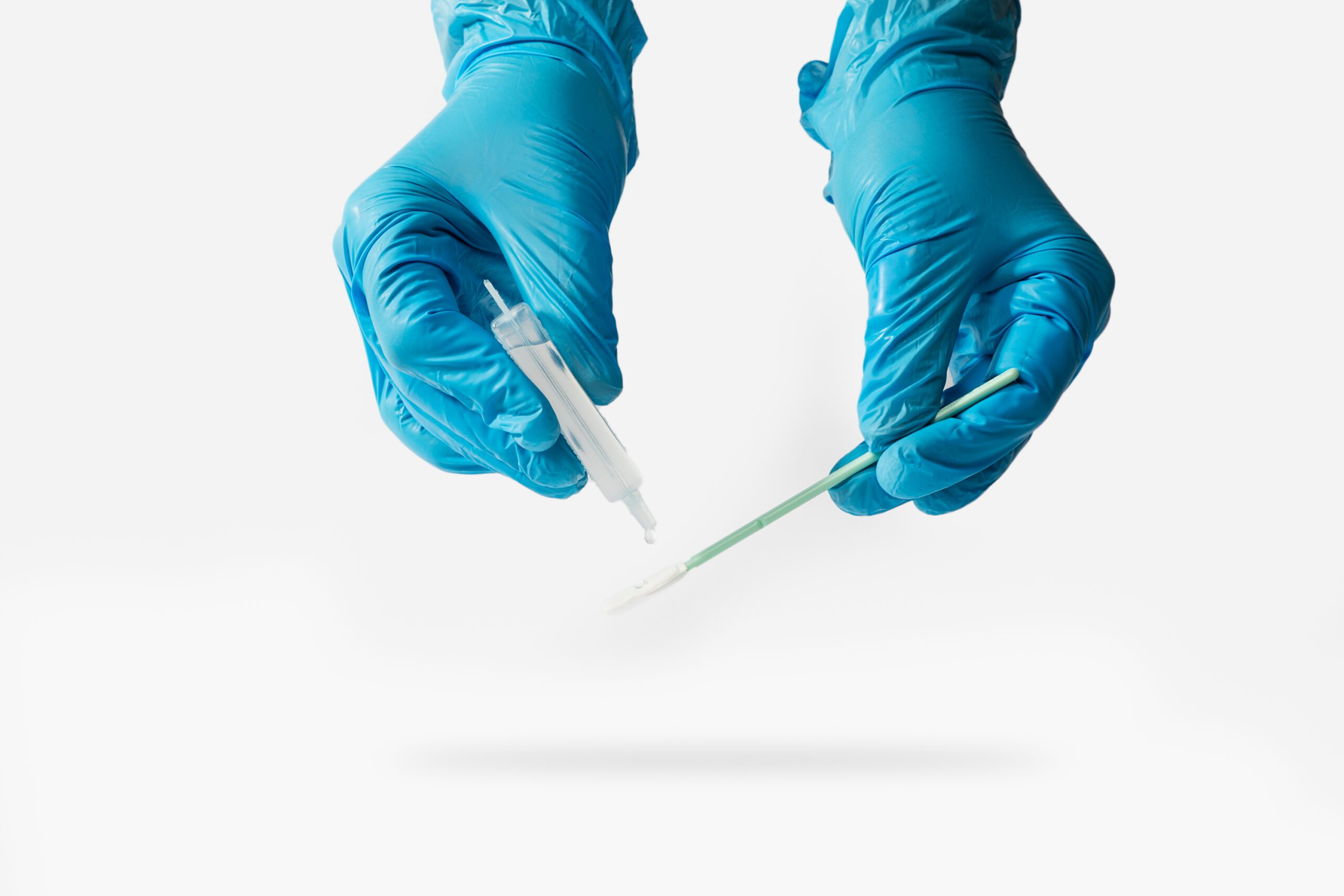
2 – Sample Sets Tube, *Texwipe ® Swab, Single-use ial of Sterile Water, Pair of Gloves in re-closable Plastic Bag, 10×10 cm (100 cm²) Template
¹The base kit can be customized according to the number of sample sets required. |
Price for Testing
Test pricing is a flat fee per Swab/Analyte/Location (ONE swab can identify ONE analyte at ONE location). No full panel needed, pay only for the analyte you need.
**CLICK HERE to download a comprehensive list of our pricing**
FAQ
Once the samples are properly collected and ready for shipping, they are placed and sealed inside the included insulated mailer. Complete the sample submission process in EagleTrax and mail the insulated mailer to Eagle for processing.
If you have any questions about the packing or shipping process or need further assistance of any kind, please contact the Eagle Care Team at 800-745-8916.
Eagle Analytical
Attn: Sample Submission
11111 S. Wilcrest Dr., S1000
Houston, Texas 77099
Click to view the Sample Submissions page which goes over the sample submission process.
Each sampling kit includes a sample instruction card, as well as a QR code that provides access to step-by-step video instruction.
We recommend using the single-use vial of sterile water that is included in the kit but deionized or distilled water may be used.
Excerpt from USP <800>:
Section 6. ENVIRONMENTAL QUALITY AND CONTROL
Environmental wipe sampling for HD surface residue should be performed routinely (e.g., initially as a benchmark and at least every 6 months, or more often as needed, to verify containment). Surface wipe sampling should include:
- Interior of the C-PEC and the equipment contained in it
- Pass-through chambers
- Surfaces in staging or work areas near the C-PEC
- Areas adjacent to C-PECs (e.g., floors directly under C-PEC, staging, and dispensing area)
- Areas immediately outside the HD buffer room or the C-SCA
- Patient administration areas
USP <800> states: “Environmental wipe sampling for HD surface residue should be performed routinely (e.g., initially as a benchmark and at least every 6 months, or more often as needed, to verify containment).”
10 cm x 10 cm (4″ x 4”)
Hormones: Estradiol, Estriol, Estrone, Ethinyl Estradiol, DHEA, Hydroxyprogesterone, Methyltestosterone, Progesterone, Testosterone, Testosterone Cypionate, Testosterone Propionate.
Chemotherapy Drugs: Carboplatin, Cisplatin, Cyclophosphamide, Cytarabine, Daunorubicin, Doxorubicin, 5-Fluorouracil, Gemcitabine, Ifosfamide, Methotrexate.
Hazardous Drugs: Apomorphine, Azathioprine, Chloramphenicol, Colchicine, Finasteride, Fluconazole, Methimazole, Oxytocin, Spironolactone, Tamoxifen Citrate, Tretinoin, Zonisamide.
Non-Hazardous Drugs: Ascorbic Acid, Tadalafil
Contact Client Care at 1-800-745-8916 ; the option of developing a method is possible.
We recommend one active per swab to ensure the highest accuracy.
Our standard turnaround is 5 business days.
*Texwipe ® is a registered trademark of Texwipe Company, LCC [Illinois Toll Works, Inc. (ITW)]
Educational Resources, Related Topics, & Events
- ????USP PUBLISHED REVISIONS – WHAT CHANGED?
- The Who, What, Where & Whys of the SurfaceShield Wipe Sampling and Cleaning Verification Kit (Product Information Page)
- Environmental Monitoring (Article)
- Proper Environmental Surface Sampling with Agar Plates (Video)
- Consulting Services & Compliance Solutions (Services)






Kanish Sangani –
This is one of the easiest test kit I have found in the market. It is simple to use and instructions are very clear and to the point. It comes with an ice pack and easy to send back in the package you receive. I would definitely recommend this service from eagle for accurate results. Understanding result is clear and their quality control staff is very friendly to help you with any question when you get the results back.
Eagle Analytical –
Thank you, Kanish. We’ve put a lot of thought into making the kit and its testing be of great value. So, we are super excited to know that you have found the kit to be an easy way to perform sampling and receive accurate results! Thank you for allowing us to assist you, sir. We are here anytime you need us.