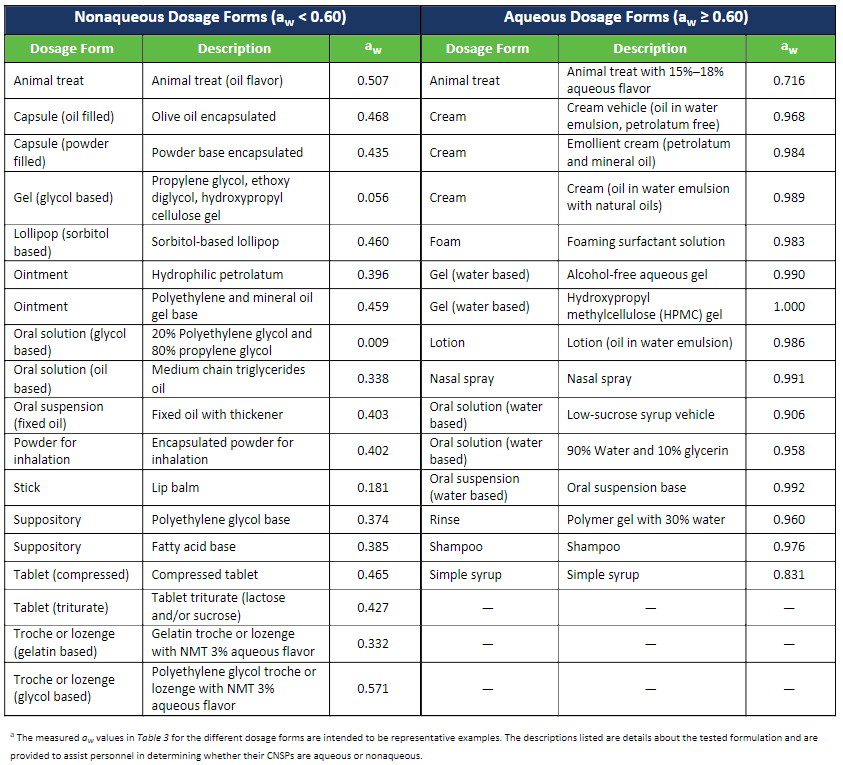
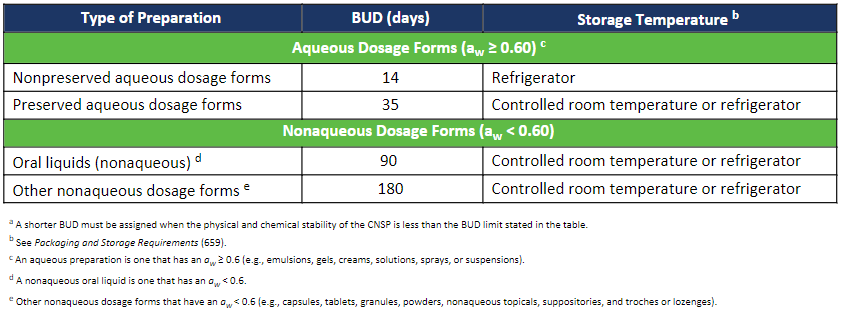
In the past, pharmacists were tasked with combing through multiple sources of literature and previous studies to determine an appropriate BUD for a specific formulation. However, revisions to USP <795> that will take effect on November 1, 2023, introduce the use of a dosage forms’ water activity as the basis for establishing a BUD for compounded nonsterile preparations. This approach has simplified the process of assigning BUDs by taking the guesswork out of the equation and eliminating the need to review multiple sources; ultimately saving the pharmacist time and effort.
The new practice of using water activity as a tool for establishing the BUD of a CNSP is described in Section 10.3 of the to-be-official USP <795> chapter. Water activity testing in accordance with USP <922> is not required but is highly recommended to support the established BUD of the CNSP. Water activity testing should be considered in cases where there is an absence of a USP-NF Compounded Preparation Monograph and/or CNSP-specific stability information.