EAGLE IS MORE THAN TESTING, LET OUR KNOWLEDGE WORK FOR YOU
GAP ANALYSIS AUDIT | COMPLIANCE SOLUTIONS & CONSULTING | CALIBRATIONS & CERTIFICATIONS | TESTING | & MORE
USP <795> & <797> Antimicrobial Effectiveness
Antimicrobial Effectiveness
Thank you for reading this post, don't forget to subscribe!
Antimicrobial preservatives are substances added to sterile and nonsterile dosage forms to inhibit the growth of microorganisms in multiple–dose products. Antimicrobial effectiveness should be demonstrated for all products containing preservatives including injections and other aqueous-based, multiple–dose products not limited to ophthalmics, topicals, and oral dosage forms.
Revisions to USP compounding chapters <795> Pharmaceutical Compounding – Nonsterile Preparations and <797> Pharmaceutical Compounding – Sterile Preparations offer more clarity regarding the requirements for conducting antimicrobial effectiveness testing (AET) and identify specific exceptions to these requirements.
AET is critical in establishing beyond-use dates (BUD) for compounded sterile preparations (CSPs) and extending BUDs for compounded nonsterile preparations (CNSPs).
Antimicrobial Effectiveness Testing in Compounded Sterile Preparations (CSPs)
Revisions to USP <797> mandate that aqueous, multiple-dose CSPs undergo AET in accordance with USP <51> once per formulation in its specific container-closure system. However, the compounder may rely on published literature or results from an FDA-registered facility if the following conditions apply:
- The formulation, both in strength and ingredients, is equivalent to the reference formulation.
- The container closure-system is equivalent to the one tested.
In lieu of testing each formulation for AET, a compounder may perform a bracketing study for any given formulation of varying strengths. This approach tests antimicrobial effectiveness in a formulation at the lowest and highest active pharmaceutical ingredient (API) concentration and the results would extend to all formulations compounded within the tested range, given that all other excipient concentrations remain the same.
For example, if a compounder formulates drug product X at a concentration of 1, 5, 10, and 15% in the same container-closure system, then AET can be performed on the 1 and 15% concentrations to satisfy the requirements of the chapter.
As with the previous version of the chapter, multiple-dose CSPs must be discarded 28 days after initial puncture unless otherwise specified by the manufacturer, but never to exceed the BUD.
Additionally, all multiple-dose CSPs must be prepared in accordance with Category 2 or 3 requirements of the to-be-official chapter.
Non-preserved aqueous and topical ophthalmic CSPs that are prepared as Category 2 or 3, labeled for single-patient use, and labeled to be discarded within 24 hours after opening at room temperature or 72 hours when refrigerated, do not require AET.
Antimicrobial Effectiveness Testing in Compounded Nonsterile Preparations (CNSPs)
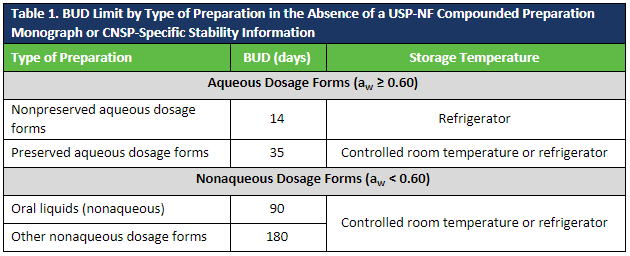
Revisions to USP <795> require AET when BUDs of preserved aqueous CNSPs are extended beyond the limits defined in Table 4 of the to-be-official chapter or Table 1 below. Additionally, for higher water activity dosage forms, AET must be performed to ensure that the antimicrobial agent is suitable to protect against proliferation of bacteria, yeast, and mold contamination that may be inadvertently introduced anytime during the compounding process, handling, and storage conditions.
To learn more about water activity, click here.
If you would like to learn more about USP <51> Antimicrobial Effectiveness Testing, contact a member of our Client Care team today!
OUR LABORATORIES
Eagle’s cutting-edge 35,000-square-foot facility and laboratory are equipped with advanced technologies and specialized segregated laboratory spaces to meet the diverse needs of our clients. This behind-the-scenes video provides you with an opportunity to witness testing while touring our laboratory.