USP Revisions
Antimicrobial Effectiveness
Revisions to USP compounding chapters <795> Pharmaceutical Compounding – Nonsterile Preparation and <797> Pharmaceutical Compounding – Sterile Preparations offer more clarity regarding the requirements for conducting antimicrobial effectiveness testing (AET) and identify specific exceptions to these requirements.
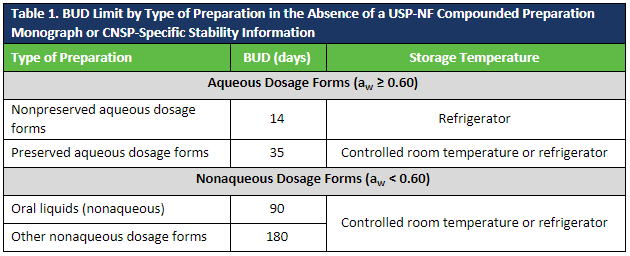
AET is critical in establishing beyond-use dates (BUD) for compounded sterile preparations (CSPs) and extending BUDs for compounded nonsterile preparations (CNSPs).