USP Revisions
Beyond-Use Dating
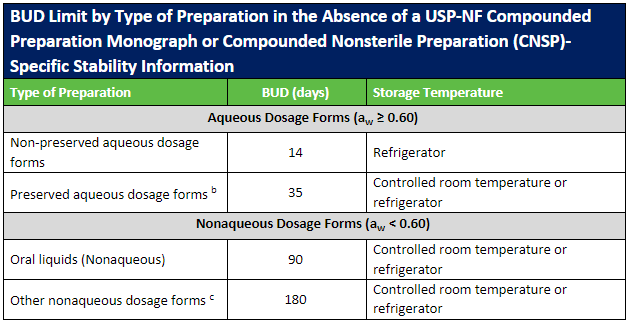
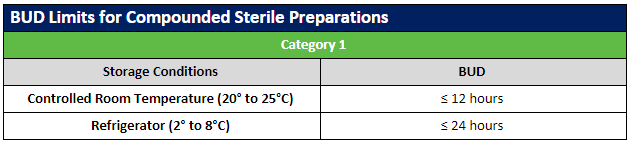
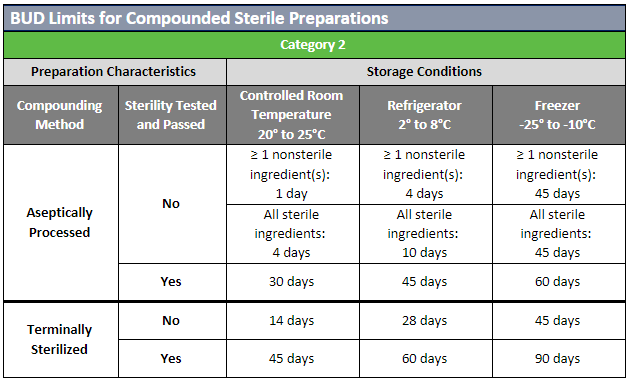
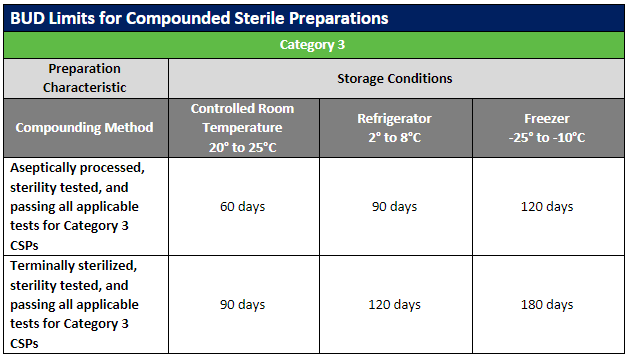
The USP published updates to the USP General Chapters on compounding nonsterile (USP 795) and sterile (USP 797) preparations. Included in these revisions were updates to the beyond-use dates (BUDs) to clarify topics that were not consistently understood. Most of the revisions involved guidance on stability and sterility considerations for nonsterile and sterile preparations.